 
Master Teachers cater to teaching Maths, Physics, Chemistry and Biology (Science) for 6th to 12th grades across CBSE and ICSE Boards. To ensure that motivation is stirred in the best proportion for your clear understanding, a good number of quizzes and Objective tests like V-Brainer, V-Maths, Turbo Maths are organized to impart knowledge and reward the best performers with surprise gifts. Hours and Hours of Study with no fun, is a bad idea for you, foreseeing the long run. Vedantu is the first choice of students aspiring to score full marks in their ICSE and CBSE Board exams or to crack any competitive exam like IIT JEE (Mains & Advanced), Kishore Vaigyanik Protsahan Yojana (KVPY), National Talent Search Exam (NTSE), International Math Olympiad (IMO), International English Olympiad (IEO). Interactive approach establishes a well-deserved academic connect between you and Master Teachers. Sessions get recorded for you to access for quick revision later, just by a quick login to your account. Your academic progress report is shared during the Parents Teachers Meeting. Assignments, Regular Homeworks, Subjective & Objective Tests promote your regular practice of the topics. Revision notes and formula sheets are shared with you, for grasping the toughest concepts. WAVE platform encourages your Online engagement with the Master Teachers. We provide you year-long structured coaching classes for CBSE and ICSE Board & JEE and NEET entrance exam preparation at affordable tuition fees, with an exclusive session for clearing doubts, ensuring that neither you nor the topics remain unattended. We have grown leaps and bounds to be the best Online Tuition Website in India with immensely talented Vedantu Master Teachers, from the most reputed institutions. Vedantu LIVE Online Master Classes is an incredibly personalized tutoring platform for you, while you are staying at your home. In the medical field, up to 5% of carbon dioxide is combined with oxygen to stimulate breath in serious conditions. 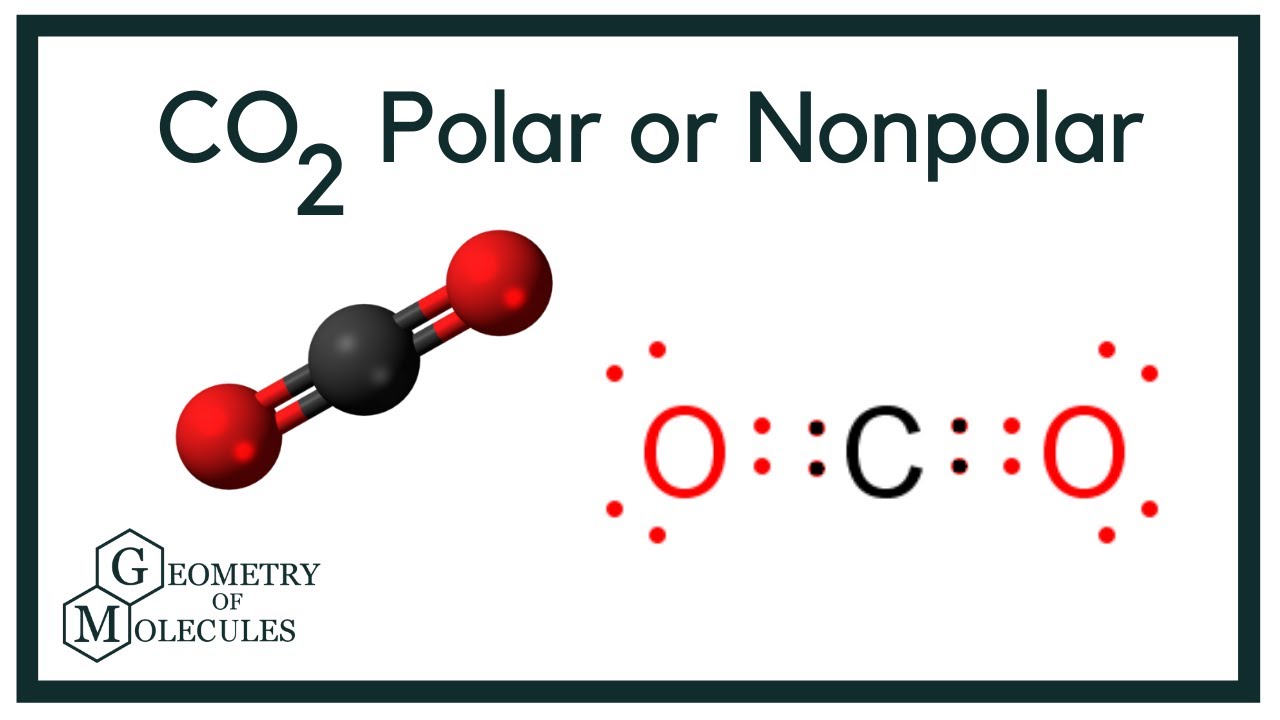
Carbon dioxide is widely used in food and oil processing companiesħ. Carbon dioxide is used to produce soda water and carbonated soft drinks.Ħ. Carbon dioxide plays an important role in photosynthesis, which is predominant for starch preparation.ĥ. Carbon dioxide is used as a fire extinguisher since it can control the spread of fire due to its non-flammable characteristics.Ĥ. Carbon dioxides are used to form dry ice, which is used for refrigeration.ģ. Carbon dioxide is used as a coolant for wastewater management systems.Ģ. Carbonic acids are formed by dissolving carbon dioxide in water.ġ. Carbonic acid is a weak acid because the ionization with water is incomplete. So, carbon dioxide is present in the rivers, lakes, groundwater, ice caps, petroleum, and natural gases. 
Carbon dioxide has an acidic odour and generates a taste like soda water in the mouth.Ĭarbon dioxide can dissolve in water and forms an aqueous solution. CO 2 is what the carbon dioxide formula is. Carbon is the sixth element present in the periodic table. Further, carbon dioxide(CO 2 ) is the fourth highest chemical present in the entire universe. Next to oxygen, carbon is abundantly found in the human body. The best know form of carbon in the atmosphere is diamond and graphite. This can establish multistable covalent bonds with suitable atoms. The carbon has the capacity to establish bonds with other atoms easily. Carbon is a tetravalent compound, which can accommodate 4 electrons to form a covalent bond. What is the Chemical Formula of Carbon Dioxide?Ĭarbon is a non-metallic chemical compound with atomic number 6. Carbon dioxide exists in hexagonal form and face-centered diamond-cubic structure. The molecular configuration of carbon dioxide is 1s 2 2s 2 2p 2. Carbon dioxide can soluble in water and forms carbonic acid. The length of the bond between carbon-oxygen is 116.3 pm. C is what the chemical formula of carbon is written as, which is the non-metallic chemical element present in Group 14 of the periodical table. Carbon dioxide is released by all the aerobic respiration. Carbon dioxide molecules consist of a carbon atom which consists of a covalently double bond with two oxygen atoms. Carbon Dioxide is a colourless acidic gas. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
